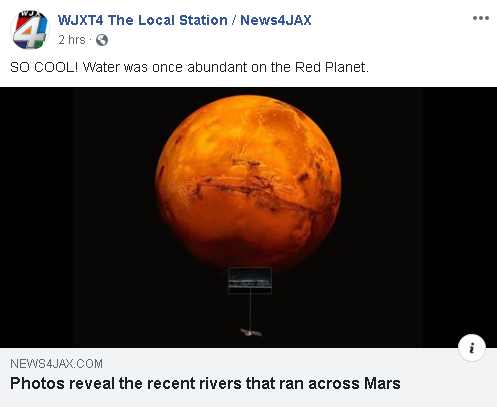
For actual H2O water to exist on a planet, there are several conditions that have to be met.
- There has to be enough “free oxygen molecules” available to bond with hydrogen molecules to create water. 98% of the atmosphere on Mars is CO2. Which means most all the oxygen molecules are already bonded to a carbon molecule which means no available oxygen to bond with hydrogen.
- There has to be enough hydrogen molecules to bond with oxygen to create water. And since the atmosphere is 98% CO2, means that there is only trace amounts of hydrogen. About enough to make a pond or a very small lake.
- The barometric pressures of the atmosphere has to be just right for the molecules to go through the phase transition of gas to liquid. Earth’s barometric pressures and temps are just right for this to happen. Mars is not. A planet’s barometric pressures also determine the boiling point of water. Earth’s is a little over 200 degrees F. But because Mar’s barometric pressures are 1/32 that of earth, water boils at about 50 degrees F. And mar’s highest temps reach about 70 degrees, if there were any oceans they would boil.
- But what about the ice we see?…. What NASA refuses to talk about is that there are 2 types of ice that can exist on Mars. H2O ice aka solid water, and CO2 ice aka dry ice aka solid CO2. Both look the same but dry ice requires colder temps to freeze. Mars has these temps in the polar regions, and with the 98% CO2 atmosphere, the CO2 would freeze in mid air and fall like H2O snow and look like it as well. And because NASA knows everyone would like to believe in other life on other planets, it’s not really hard to sell the idea if you ignore the science. and outright lie.
- So what about the areas that show liquid flow evidence? 2 things. CO2 has a liquid state that can flow. And lava flows. Flow erosion does not always equal water existed or every planet would have it. There is more than water that can cause erosion evidence.
- But what about that liquid that one of the mobile vehicles ran into on Mars? That was CO2 melting, and that which was seen was CO2 in it’s liquid state. CO2 frozen goes from solid to liquid to gas just like frozen water does. The rover ran upon CO2 in it’s liquid state.
If you disagree with this then you need to explain how something is made without the molecules that are it’s basic makeup?